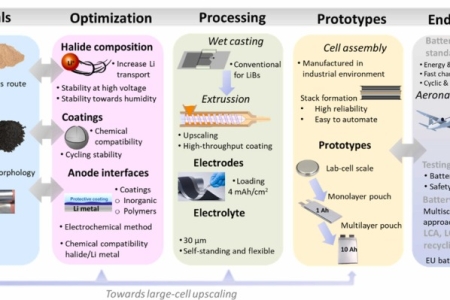
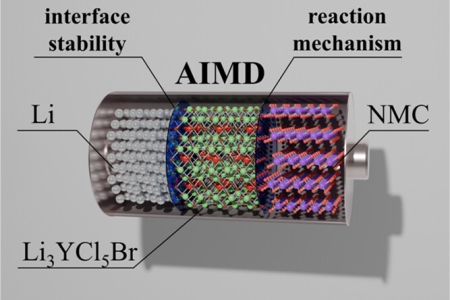
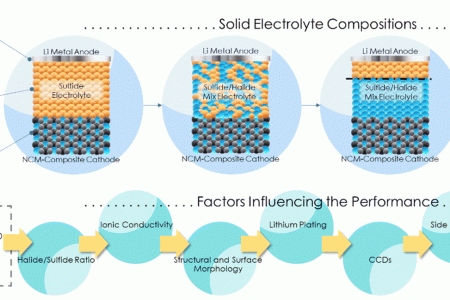
Stable cycling of halide solid state electrolyte enabled by a dynamic layered solid electrolyte interphase between Li metal and Li3YCl4Br2
Halides are potential electrolytes for Li metal solid state batteries owing to their combination of high ionic conductivity, ductility and electrochemical stability against oxidation. However, their reactivity with the Li metal electrode may result in the formation of secondary compounds hindering their practical utility in terms of cycling performance as key indicator in battery operation. In this work, we investigate the high performance of symmetric cells with Li3YCl4Br2 halide and bare Li-metal electrode, able to withstand 1000 h of Li electrodeposition-dissolution with an overpotential as low as 46 mV. Through a comprehensive analysis employing physico-chemical and electrochemical characterizations, complemented by computational methodologies, we unravel the dynamics of the complex of the Li/halide interface and its evolution during cycling. The reactivity between Li3YCl4Br2 with metallic Li results in the reduction of the halide into LiCl, LiBr and Y metal. Surprisingly, during cycling, those secondary products from the reduction of the halide build a structured solid electrolyte interphase, containing a Y-rich electronic conductive and LiCl and LiBr ionic conductive layers. The particular chemistry and robustness of this solid electrolyte interphase exhibiting a mixed ionic and electronic conductivity appears to be responsible for the outstanding cycling stability.